Search results for: 'instant view lh ovulation predicting test cassett'
-
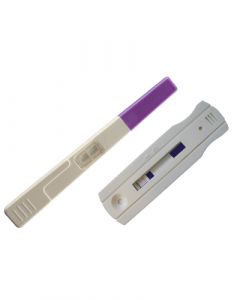 Instant View LH Ovulation Predicting Tests (Cassette)SKU :ALF-02-2542(25 Tests) Ovulation Predictor Test (Cassette) Kits, the Instant View LH urine test cassette is a qualitative lateral f...$29.95
Instant View LH Ovulation Predicting Tests (Cassette)SKU :ALF-02-2542(25 Tests) Ovulation Predictor Test (Cassette) Kits, the Instant View LH urine test cassette is a qualitative lateral f...$29.95 -
 ACON LH Ovulation Urine Test CassetteSKU :FLH-102Item is discontinued. See below for alternative product: Instant View LH Ovulation Predicting Test (cliawaived.com...
ACON LH Ovulation Urine Test CassetteSKU :FLH-102Item is discontinued. See below for alternative product: Instant View LH Ovulation Predicting Test (cliawaived.com... -
 Instant-View Combo (Urine/Serum) CassettesSKU :ALF-02-2483(25 Tests) The Instant-View Pregnancy Combo (Urine/Serum) Test Cassette, is a qualitative immunoassay for the detection...$25.90
Instant-View Combo (Urine/Serum) CassettesSKU :ALF-02-2483(25 Tests) The Instant-View Pregnancy Combo (Urine/Serum) Test Cassette, is a qualitative immunoassay for the detection...$25.90 -
 Instant-View Morphine Drug TestsSKU :ALF-03-3093(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70
Instant-View Morphine Drug TestsSKU :ALF-03-3093(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70 -
 Instant-View Methamphetamine Drug TestsSKU :ALF-03-3032(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$22.50
Instant-View Methamphetamine Drug TestsSKU :ALF-03-3032(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$22.50 -
 Instant-View Amphetamine Drug TestsSKU :ALF-03-3012(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70
Instant-View Amphetamine Drug TestsSKU :ALF-03-3012(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70 -
 Instant-View Propoxyphene Drug TestsSKU :ALF-03-2932(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$25.90
Instant-View Propoxyphene Drug TestsSKU :ALF-03-2932(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$25.90 -
 Instant-View Methadone Drug TestsSKU :ALF-03-3172(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70
Instant-View Methadone Drug TestsSKU :ALF-03-3172(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70 -
 Instant-View Pregnancy Urine Test CardsSKU :ALF-02-2482(25 Tests) Out of Stock! See below suggested alternative manufactured by ALFA Scientific and is the same exact HCG tes...
Instant-View Pregnancy Urine Test CardsSKU :ALF-02-2482(25 Tests) Out of Stock! See below suggested alternative manufactured by ALFA Scientific and is the same exact HCG tes... -
 Instant-View PSA Whole Blood TestSKU :ALF-05-7771(25 Tests) The Instant View PSA (Prostate Specific Antigen) semi quantitative whole blood rapid test is designed to det...
Instant-View PSA Whole Blood TestSKU :ALF-05-7771(25 Tests) The Instant View PSA (Prostate Specific Antigen) semi quantitative whole blood rapid test is designed to det... -
 Instant-View Whole Blood Troponin I TestsSKU :ALF-05-8575(25 Tests) Instant-View One Step Rapid Test for Troponin I (TnI) (Whole Blood or Serum). Rapid immunoassay as an aid in...$75.00
Instant-View Whole Blood Troponin I TestsSKU :ALF-05-8575(25 Tests) Instant-View One Step Rapid Test for Troponin I (TnI) (Whole Blood or Serum). Rapid immunoassay as an aid in...$75.00 -
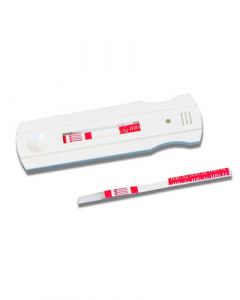 Instant View H. pylori Whole Blood Rapid TestSKU :ALF-05-9475(25 Tests) INSTANT VIEW H. PYLORI WHOLE BLOOD RAPID TEST. This FDA 510K cleared, CLIA Waived, and CE marked test is a r...$62.00
Instant View H. pylori Whole Blood Rapid TestSKU :ALF-05-9475(25 Tests) INSTANT VIEW H. PYLORI WHOLE BLOOD RAPID TEST. This FDA 510K cleared, CLIA Waived, and CE marked test is a r...$62.00 -
 Instant-View Methamphetamine Drug TestsSKU :ALF-03-3033(25 Tests) Methamphetamine 500 MET Drug Screening Kits, rapid onsite drug test for detection of drugs of abuse in urine...$20.70
Instant-View Methamphetamine Drug TestsSKU :ALF-03-3033(25 Tests) Methamphetamine 500 MET Drug Screening Kits, rapid onsite drug test for detection of drugs of abuse in urine...$20.70 -
 Instant-View Cocaine Drug TestsSKU :ALF-03-3052(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70
Instant-View Cocaine Drug TestsSKU :ALF-03-3052(25 Tests) Rapid onsite drug test for detection of drugs of abuse in urine. Simply dip card into specimen cup or apply ...$20.70 -
 Instant-View 9-Panel Drug TestsSKU :ALF-03-3997(20 Tests) 9-Panel Instant-View drug testing card, tests for: THC, COC, MOR, AMP, PCP, MET, BZD, BAR, MTD(Cup not inclu...$155.25
Instant-View 9-Panel Drug TestsSKU :ALF-03-3997(20 Tests) 9-Panel Instant-View drug testing card, tests for: THC, COC, MOR, AMP, PCP, MET, BZD, BAR, MTD(Cup not inclu...$155.25 -
 Instant-View 12-Panel Drug TestsSKU :ALF-03-3958(20 Tests) Rapid 12-panel onsite drug testing card, for detection of THC, COC, MOR300, AMP, PCP, mAMP,BZD, BAR, MTD, TC...$207.00
Instant-View 12-Panel Drug TestsSKU :ALF-03-3958(20 Tests) Rapid 12-panel onsite drug testing card, for detection of THC, COC, MOR300, AMP, PCP, mAMP,BZD, BAR, MTD, TC...$207.00 -
 Instant-View 10-Panel Drug TestsSKU :ALF-03-3917(20 Tests) Rapid 10-panel onsite drug testing card, for detection of THC, COC, MOR300, AMP, PCP, mAMP, BZD, BAR, MTD, X...$165.60
Instant-View 10-Panel Drug TestsSKU :ALF-03-3917(20 Tests) Rapid 10-panel onsite drug testing card, for detection of THC, COC, MOR300, AMP, PCP, mAMP, BZD, BAR, MTD, X...$165.60 -
 Instant-View 10-Panel Drug TestsSKU :ALF-03-3910(20 Tests) Rapid 10-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP, mAMP,BZD, BAR, MTD, TCA i...$165.60
Instant-View 10-Panel Drug TestsSKU :ALF-03-3910(20 Tests) Rapid 10-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP, mAMP,BZD, BAR, MTD, TCA i...$165.60 -
 Instant-View 6-Panel Drug TestsSKU :ALF-03-3669(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR 300, BAR, BZD, AMP in urine. (Cup not...$119.00
Instant-View 6-Panel Drug TestsSKU :ALF-03-3669(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR 300, BAR, BZD, AMP in urine. (Cup not...$119.00 -
 Instant-View 6-Panel Drug Test CardsSKU :ALF-03-3635(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, mAMP, and BZO in urine. (Cup no...$119.00
Instant-View 6-Panel Drug Test CardsSKU :ALF-03-3635(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, mAMP, and BZO in urine. (Cup no...$119.00 -
 Instant-View 6-Panel Drug TestsSKU :ALF-03-3630(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP, mAMP in urine. (Cup not in...$119.00
Instant-View 6-Panel Drug TestsSKU :ALF-03-3630(25 Tests) Rapid 6-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP, mAMP in urine. (Cup not in...$119.00 -
 Instant-View 5-Panel Drug TestsSKU :ALF-03-3577(25 Tests) Rapid 5-panel onsite drug testing card, for detection of THC/COC/MOR/mAMP/AMP in urine. (Cup not included) F...$98.30
Instant-View 5-Panel Drug TestsSKU :ALF-03-3577(25 Tests) Rapid 5-panel onsite drug testing card, for detection of THC/COC/MOR/mAMP/AMP in urine. (Cup not included) F...$98.30 -
 Instant-View 5-Panel Drug TestsSKU :ALF-03-3576QWRapid 5-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP in urine. (Cup not included) FDA-Cleare...$98.30
Instant-View 5-Panel Drug TestsSKU :ALF-03-3576QWRapid 5-panel onsite drug testing card, for detection of THC, COC, MOR, AMP, PCP in urine. (Cup not included) FDA-Cleare...$98.30 -
 Instant-View 5-Panel Drug TestsSKU :ALF-03-3571(25 Tests) Rapid 5-panel drug testing card, for detection of THC, COC, MOR, mAMP, BZD in urine. (Cup not included) FDA-...$98.30
Instant-View 5-Panel Drug TestsSKU :ALF-03-3571(25 Tests) Rapid 5-panel drug testing card, for detection of THC, COC, MOR, mAMP, BZD in urine. (Cup not included) FDA-...$98.30 -
 Instant-View 3-Panel Drug TestsSKU :ALF-03-3375(25 Tests) Rapid onsite 3-panel drug testing card for detection of THC, COC, AMP in urine. Cup not included. FDA-Cleare...$62.10
Instant-View 3-Panel Drug TestsSKU :ALF-03-3375(25 Tests) Rapid onsite 3-panel drug testing card for detection of THC, COC, AMP in urine. Cup not included. FDA-Cleare...$62.10 -
 Instant-view DF iFOB TestSKU :ALF-05-6732OTNow in stock! Expiration Date of 9/2025! First ever at-home iFOB Test! New Tiered Pricing Quantity Required ...$8.99
Instant-view DF iFOB TestSKU :ALF-05-6732OTNow in stock! Expiration Date of 9/2025! First ever at-home iFOB Test! New Tiered Pricing Quantity Required ...$8.99 -
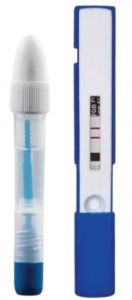 Instant-View Immunochemical Fecal Occult Blood TestSKU :ALF-05-7632(25 tests) The Instant-view plus Immunochemical Fecal Occult Blood Test is a qualitative immunoassay for detection of F...$74.75
Instant-View Immunochemical Fecal Occult Blood TestSKU :ALF-05-7632(25 tests) The Instant-view plus Immunochemical Fecal Occult Blood Test is a qualitative immunoassay for detection of F...$74.75 -
 Instant-View Combo (Urine/Serum) Dip StripsSKU :ALF-02-2488(50 Tests) The Instant-View Pregnancy Combo (Urine/Serum) Dip Strip, is a qualitative immunoassay for the detection of ...$31.05
Instant-View Combo (Urine/Serum) Dip StripsSKU :ALF-02-2488(50 Tests) The Instant-View Pregnancy Combo (Urine/Serum) Dip Strip, is a qualitative immunoassay for the detection of ...$31.05 -
 Instant-View Pregnancy Urine Dip StripsSKU :ALF-02-2487(50 Tests) This item is unavailable, consider our substitute item CLIA-02-2470. The Instant-View Pregnancy Urine ...$47.21
Instant-View Pregnancy Urine Dip StripsSKU :ALF-02-2487(50 Tests) This item is unavailable, consider our substitute item CLIA-02-2470. The Instant-View Pregnancy Urine ...$47.21 -
 Instant-View 9-Panel Drug TestsSKU :ALF-03-3989(20 Tests) 9-Panel Instant-View drug test card, tests for: AMP, BAR, BZD, COC, MET 100, MOR 300, MTD, PCP, THC. (Cup no...
Instant-View 9-Panel Drug TestsSKU :ALF-03-3989(20 Tests) 9-Panel Instant-View drug test card, tests for: AMP, BAR, BZD, COC, MET 100, MOR 300, MTD, PCP, THC. (Cup no... -
 Instant-View 12-Panel Drug Test CardsSKU :ALF-03-3965QW(20 Tests) Rapid 12-panel onsite drug testing card, for detection of Test for THC, COC, MOR 300, AMP, BAR, BZD, MET, MT...$199.00
Instant-View 12-Panel Drug Test CardsSKU :ALF-03-3965QW(20 Tests) Rapid 12-panel onsite drug testing card, for detection of Test for THC, COC, MOR 300, AMP, BAR, BZD, MET, MT...$199.00 -
 Instant View Single Panel AmphetamineSKU :ALF-03-3014Instant View Single Panel Amphetamine 300 (25 tests)...$22.50
Instant View Single Panel AmphetamineSKU :ALF-03-3014Instant View Single Panel Amphetamine 300 (25 tests)...$22.50 -
 CLIAwaived, Inc. Pregnancy Tests (50 Cassettes)SKU :CLIA-02-2479(50 Tests) The CLIAwaived,Inc. Pregnancy Test (Cassette) test, 7mm hCG Top, is a rapid one step visual test for the qua...$39.35
CLIAwaived, Inc. Pregnancy Tests (50 Cassettes)SKU :CLIA-02-2479(50 Tests) The CLIAwaived,Inc. Pregnancy Test (Cassette) test, 7mm hCG Top, is a rapid one step visual test for the qua...$39.35 -
 CLIAwaived, Inc. Pregnancy Tests (25 Cassettes)SKU :CLIA-02-2478(25 Tests) The CLIAwaived, Inc. Pregnancy Test (Cassette), 7mm hCG Top, is a rapid one step visual test for the qualita...$18.99
CLIAwaived, Inc. Pregnancy Tests (25 Cassettes)SKU :CLIA-02-2478(25 Tests) The CLIAwaived, Inc. Pregnancy Test (Cassette), 7mm hCG Top, is a rapid one step visual test for the qualita...$18.99 -
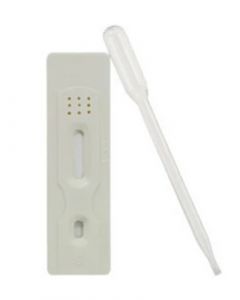 SmokeCheck Nicotine Test CassetteSKU :DCT-102(40 Tests) SmokeCheck is a simple one step urine test card for detection of tobacco or nicotine usage. SmokeCheck test ...$48.00
SmokeCheck Nicotine Test CassetteSKU :DCT-102(40 Tests) SmokeCheck is a simple one step urine test card for detection of tobacco or nicotine usage. SmokeCheck test ...$48.00 -
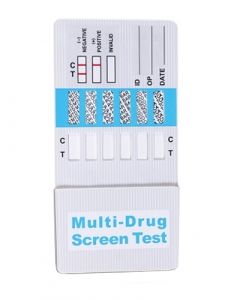 ACON 6 Panel Drug Testing CassettesSKU :DOA-2165(25 Tests) 6 Panel Drug Testing, FDA Cleared drug testing card for detection of Marijuana, Cocaine, Methamphetamine, Am...$139.75
ACON 6 Panel Drug Testing CassettesSKU :DOA-2165(25 Tests) 6 Panel Drug Testing, FDA Cleared drug testing card for detection of Marijuana, Cocaine, Methamphetamine, Am...$139.75 -
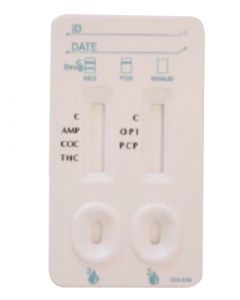 ACON 5 Panel Drug Testing CassettesSKU :DOA-3155(25 Tests) 5 Panel Drugs of Abuse Test, FDA Cleared and CLIA Waived drug testing card for detection of Marijuana, Cocai...$85.00
ACON 5 Panel Drug Testing CassettesSKU :DOA-3155(25 Tests) 5 Panel Drugs of Abuse Test, FDA Cleared and CLIA Waived drug testing card for detection of Marijuana, Cocai...$85.00 -
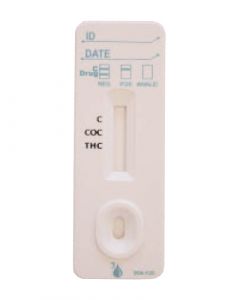 ACON 2 Panel Drug Testing CassettesSKU :DOA-1125(25 Tests) 2 Panel Drug Test, FDA Cleared and CLIA Waived drug testing card for detection of Marijuana and Cocaine in u...$58.25
ACON 2 Panel Drug Testing CassettesSKU :DOA-1125(25 Tests) 2 Panel Drug Test, FDA Cleared and CLIA Waived drug testing card for detection of Marijuana and Cocaine in u...$58.25 -
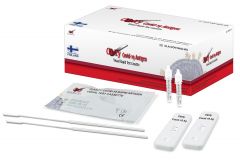 Clarity COVID-19 Nasopharyngeal Antigen Rapid Test CassettesSKU :CLA-COV19AG-VIS(25 tests) Out of Stock - see below for alternative testing options: INDICAID™ COVID-19 Rapid Antigen Test Kit (cl...
Clarity COVID-19 Nasopharyngeal Antigen Rapid Test CassettesSKU :CLA-COV19AG-VIS(25 tests) Out of Stock - see below for alternative testing options: INDICAID™ COVID-19 Rapid Antigen Test Kit (cl... -
 Malaria P.f. / P.v. / Pan Rapid Test CassettesSKU :AT-PFPVPN-402-CARD(25 Tests per Kit) Minimum order of 1000 tests! The Malaria P.f./P.v. Rapid Test Cassette (Whole Blood) is a rapid chr...
Malaria P.f. / P.v. / Pan Rapid Test CassettesSKU :AT-PFPVPN-402-CARD(25 Tests per Kit) Minimum order of 1000 tests! The Malaria P.f./P.v. Rapid Test Cassette (Whole Blood) is a rapid chr... -
 Pregnancy Urine Cassette TestsSKU :CLIA-02-2472(25 Tests) The CLIAwaived,Inc. Pregnancy Test (Cassette) test, 7mm hCG Top, is a rapid one step visual test for the qua...$21.70
Pregnancy Urine Cassette TestsSKU :CLIA-02-2472(25 Tests) The CLIAwaived,Inc. Pregnancy Test (Cassette) test, 7mm hCG Top, is a rapid one step visual test for the qua...$21.70 -
 IDTC-II 9 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-09-14Qty Left :21(25 Tests/Box) Limited Short Dated Stock! Expires 6/1/2024! For Longer Dated Stock please email [email protected]...Special Price $37.50 Regular Price $62.50
IDTC-II 9 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-09-14Qty Left :21(25 Tests/Box) Limited Short Dated Stock! Expires 6/1/2024! For Longer Dated Stock please email [email protected]...Special Price $37.50 Regular Price $62.50 -
 IDTC-II 6 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-06-264(25 Tests/Box) Tests for AMP500 / BZO300 / COC150 / MET500 / OPI (MOR 300) / THC50 The newly designed IDTC-II dip card...$62.50
IDTC-II 6 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-06-264(25 Tests/Box) Tests for AMP500 / BZO300 / COC150 / MET500 / OPI (MOR 300) / THC50 The newly designed IDTC-II dip card...$62.50 -
 IDTC-II 5 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-05-3571(25 Tests/Box) Tests for BZO300 / COC150 / MET500 / OPI (MOR 300) / THC50 The newly designed IDTC-II dip card feature...$49.75
IDTC-II 5 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-05-3571(25 Tests/Box) Tests for BZO300 / COC150 / MET500 / OPI (MOR 300) / THC50 The newly designed IDTC-II dip card feature...$49.75 -
 IDTC-II 9 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-M-09-23Item unavailable, please click Request a Quote to inquire about a substitute item, or see Related Products. (25 Tests/B...
IDTC-II 9 Panel Instant Drug Test CardsSKU :CLIA-IDTC-II-M-09-23Item unavailable, please click Request a Quote to inquire about a substitute item, or see Related Products. (25 Tests/B... -
 IDTC-II 14 Panel Instant Drug Test Cards (With BUP)SKU :CLIA-IDTC-II-14-01(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / EDDP300 / MDMA500 / MET500 / MTD300 / OPI (MOR 300...$137.50
IDTC-II 14 Panel Instant Drug Test Cards (With BUP)SKU :CLIA-IDTC-II-14-01(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / EDDP300 / MDMA500 / MET500 / MTD300 / OPI (MOR 300...$137.50 -
 IDTC-II 12 Panel Instant Drug Test Cards (With BUP & ADULT)SKU :CLIA-IDTC-II-88-A(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / MDMA500 / MET500 / MTD300 / OPI300 / OXY100 / PCP...$131.25
IDTC-II 12 Panel Instant Drug Test Cards (With BUP & ADULT)SKU :CLIA-IDTC-II-88-A(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / MDMA500 / MET500 / MTD300 / OPI300 / OXY100 / PCP...$131.25 -
 IDTC-II 12 Panel Instant Drug Test Cards (With BUP)SKU :CLIA-IDTC-II-88(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / MDMA500 / MET500 / MTD300 / OPI (MOR 300) / OXY10...$124.95
IDTC-II 12 Panel Instant Drug Test Cards (With BUP)SKU :CLIA-IDTC-II-88(25 Tests/Box) Tests for AMP500 / BAR300 / BUP10 / BZO300 / COC150 / MDMA500 / MET500 / MTD300 / OPI (MOR 300) / OXY10...$124.95

